Monitoring
Case studies Management Of GMP Data and Alarms
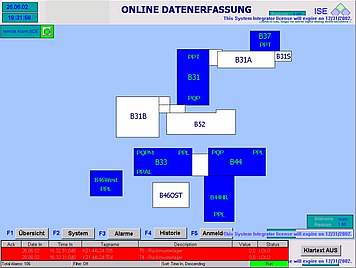
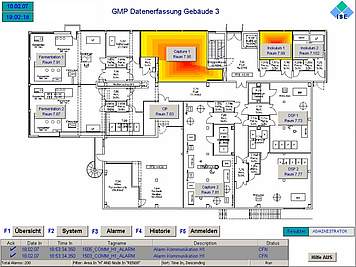
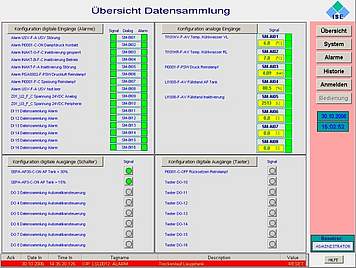
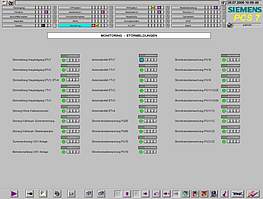
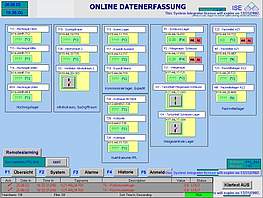
Collection, monitoring, archiving and presentation of distributed cGMP relevant data and alarm signals are some of the most important expectations towards any supervisory process control system.
We at ISE boast of vast detail knowledge about process automation in all fields of pharmaceutical industry.
We can design and configure cGMP compliant data monitoring and central alarm management in accordance with your process needs. The key requirement for this task is the integration of different process systems and package units with their specific interfaces into one uniform monitoring system, which processes collected data and makes them accessible at distributed HMI stations and allows reporting at different printing stations.
Since we are specialists in the automation of packages (bioreactors, media and buffer preparation, CIP, clean utility preparation and distribution,...) we are familiar with their interfaces.
At ISE we can help you collect data from various sources of your organization and convert them into useful information available at a click of a button. Our cGMP compliant data monitoring and alarm management systems are designed on the basis of and built up from standardised industrial components, which will facilitate faster adaption and easy maintenance.
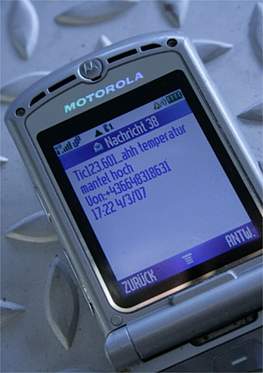
We adapt our automation systems for GMP data monitoring and alarm management to your scope and requirements, ranging from alarm relays and SCADA systems up to full-scale process control systems. All commonly used interfaces for signal and data exchange are available. Custom made data collection tools can also be designed on request or need basis. Tools for alarm and event routing to SMS and email complete our deliverables.
ISE application software is developed on standard industrial components and platforms:
PLC level:
Siemens Simatic, Allen Bradley, B&R
SCADA:
Siemens WinCC, WinCC flexible, iFix, InTouch
PCS/DCS:
Siemens PCS 7
Interfaces:
- Interfaces to Enterprise Resource Planning ERP:
e.g. SAP, Microsoft Dynamics, Oracle, Globemanager - Interfaces to Manufacturing Execution Systems MES:
e.g. WERUM PAS X, Sail - Interfaces to Plant Information Management Systems PIMS:
e.g. OSI PI, Acron, Apen Tech, iHistorian - Interfaces to Laboratory Information Management Systems LIMS:
e.g. Agilent SLIMS
Custom specific system requirement on demand.