Fermentation
Case studies Automation of Fermentation - Bioreactors - Biotechnology
ISE has been present in Life Science Industry providing state-of-the-art compliant automation solutions for more than 20 years.
Our vast experience and the demanding user requirements of our clients have helped us in developing and establishing pre-configured and standardised technological and functional modules that are easily assembled to specific process requirements. The basic design and implementation of control systems for bioprocess unit operations has been proven and validated at hundreds of application installations.
This constitutes the basis of our confidence to provide compliant automation solutions for any biotechnological process (of various scales or types).
Let us integrate this know-how into Your installation following Your specific requirements.
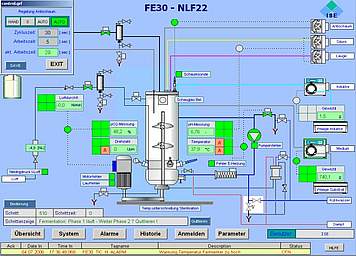
ISE automation system FE10
Your automation solution from laboratory scale to clinical trial scale for reactors with working volumes up to 30 litres
ISE automation system FE100
Your automation solution for pilot and production scale up to 150 litre reactors
ISE automation system FE1000
Your automation solution for production scale beyond 150 litre reactors
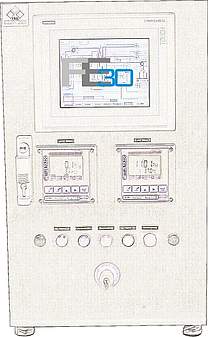
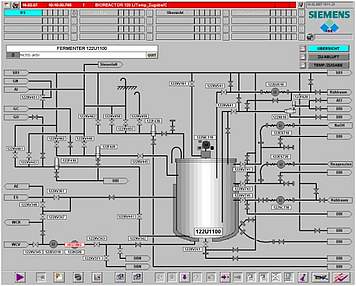
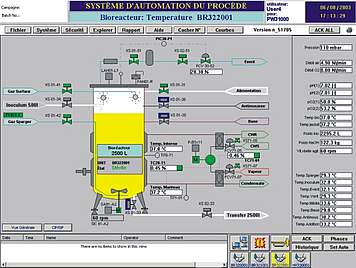
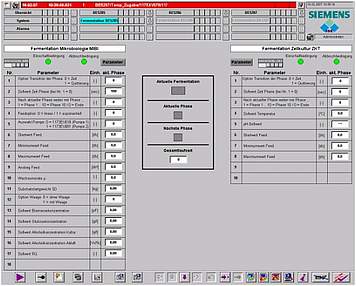
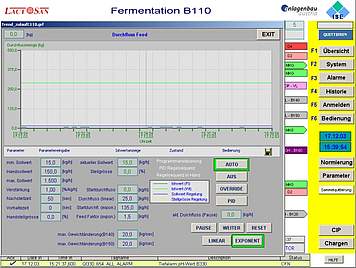
The basic design of ISE's standard automation package is based on industrial PLC systems, which perform I/O and alarm handling, smart control loops and run-off control. The HMI can be scaled from simple operator panels up to SCADA systems as well as be integrated into total plant process control systems.
Our application software runs on widely accepted industrial hardware platforms:
PLC level: Siemens Simatic, Allen Bradley, B&R
SCADA: Siemens WinCC, WinCC flexible, iFix, InTouch
PCS/DCS: Siemens PCS7
Custom specific system requirement on demand.
We offer You communication to all known interfaces on networks (TCP/IP, Industrial Ethernet,...) and field bus (profibus, CAN, foundation fieldbus, profinet).
In order to achieve regulatory compliance for our clients, we implement our applications and projects in strict accordance with the GAMP guideline's life cycle approach, which is reflected in all steps of our quality management system.
In accordance with the clients´ needs, we deliver automation system solutions which are CFR 21 part 11 compliant.
We conceive smart automation setups for standard configurations as well as for customised solutions. Besides functionality and quality we care for a design which is nice to look at and to be proud of. User friendly operator interfaces allow you to fully implement your technological know-how in your installation. Reporting, monitoring, security, data integrity and interfaces to cGMP compliant data monitoring are common parts of our automation design.